Allotropes of Carbon
Allotropes of Carbon:
What is an allotrope?
- Each of two or more different physical forms in which an element can exist. Graphite, charcoal, and diamond are all allotropes of carbon.
This page we will be discussing Carbons allotropes and providing some of the key information about those allotropes. Carbon has 8 allotropes, however, its most comm on allotropes are listed below:
Diamond,
Graphite,
Amorphous carbon,
White carbon,
Amorphous
Amorphous carbon, molecules are not considered as major allotropes of carbon. This is because they are made up of small crystals of graphite. Also, amorphous carbons are made in various fake ways. They make things like fake watches, fake diamonds, basically anything expensive that make artificially using amorphous carbon. Charcoal, soot and carbon fibres are examples of amorphous carbon.
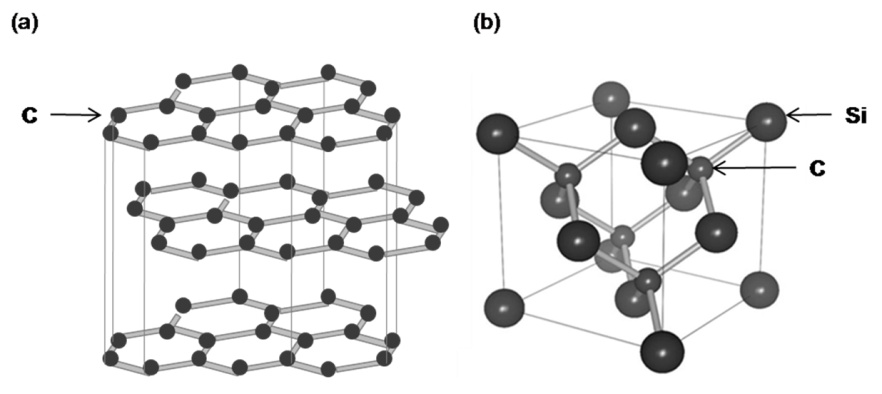
Graphite
One of the softest materials known, is a form of carbon that is primarily used as a lubricant. Naturally occurring graphite occurs in two forms, alpha and beta. These two forms have identical physical properties but different crystal structures.
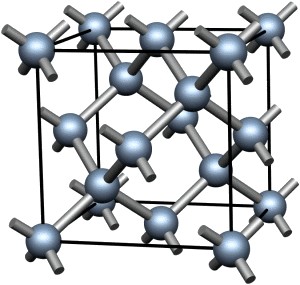
Diamond
The third naturally occurring form of carbon, is one of the hardest substances known, diamond. Naturally occurring diamond is typically used for jewellery, however, many diamonds are artificially produced. These small diamonds are made by squeezing graphite under high temperatures and pressures for days or weeks. Although, they possess very different physical properties, graphite and diamond differ only in their crystal structure.
White Carbon
A fourth allotrope of carbon, known as white carbon, was produced in 1969. It is a transparent material that can split a single beam of light into two beams. Very little is known about this form of carbon.
In the first image below it shows graphite in alpha and beta form.
In the second image below, it displays a modal of carbons most common allotrope, graphite.
The third image is a chemical structure of diamond.
Whats your favourite form of carbon?
a) graphite
b) diamond
c) amorphous carbon
d) the rare white carbon
a) graphite
b) diamond
c) amorphous carbon
d) the rare white carbon