Isotopes of Carbon
Isotopes:
Let's say an atom is missing a neutron or has an extra neutron. That type of atom is called an isotope. An atom is still the same element if it is missing an electron. The same goes for isotopes. They are still the same element. They are just a little different from every other atom of the same element.
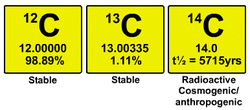
Carbon has few isotopes. All of which are listed in the two images above.
Carbon 14, is no very abundant and is not stable. It is radioactive and cosmogenic. Carbon 14 has 6 protons, 6 electrons and 8 neutrons.
Carbon 13, is the second most abundant carbon isotope (1.11%) and is stable. Carbon 13 also has 6 protons, 6 electrons but 7 neutrons.
Carbon 12, is the most abundant carbon isotope (98.89%) and is very stable. Carbon 12 has 6 electrons and protons, as well as 6 neutrons.
The reason why Carbons atomic mass is 12.011 is because of the isotopes. take a look here:
Carbon 12 (98.89%)
Carbon 13 (1.11%)
99/100 x 12 + 1/100 x 13
= 11.88 + = .13
= 12.011
A piece of carbon is displayed in the image below.
Let's say an atom is missing a neutron or has an extra neutron. That type of atom is called an isotope. An atom is still the same element if it is missing an electron. The same goes for isotopes. They are still the same element. They are just a little different from every other atom of the same element.
Carbon has few isotopes. All of which are listed in the two images above.
Carbon 14, is no very abundant and is not stable. It is radioactive and cosmogenic. Carbon 14 has 6 protons, 6 electrons and 8 neutrons.
Carbon 13, is the second most abundant carbon isotope (1.11%) and is stable. Carbon 13 also has 6 protons, 6 electrons but 7 neutrons.
Carbon 12, is the most abundant carbon isotope (98.89%) and is very stable. Carbon 12 has 6 electrons and protons, as well as 6 neutrons.
The reason why Carbons atomic mass is 12.011 is because of the isotopes. take a look here:
Carbon 12 (98.89%)
Carbon 13 (1.11%)
99/100 x 12 + 1/100 x 13
= 11.88 + = .13
= 12.011
A piece of carbon is displayed in the image below.



