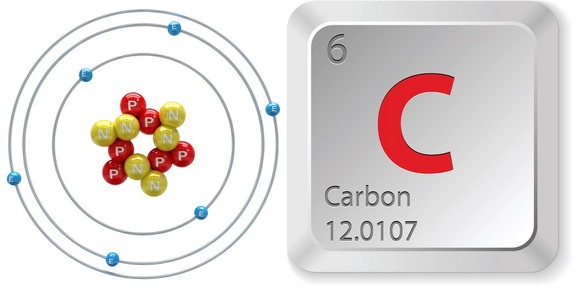
Structure
The images above and below display the structure of carbon. In both images it shows Carbon has 6 Neutrons (N) and 6 protons (P), it also shows that Carbon has 6 electrons (E) in its electron shells.
Carbon has 2 electrons in its first electron shell and in its valence electron shell (outer most shell) it has 4 electrons.
Carbon is unique in its ability to form a close to ten million different compounds. Carbon forms a stable structure with itself. Graphite is a Non-metallic, crystalline form. Diamonds are a Glossy, crystalline form.
Facts:
Ionization Energy: 11.260 eV
Oxidation States: +4, +2, -4
Electron Shell Configuration:
1s2
2s2 2p2
The slide show below is a series of images displaying carbons forms in their structures.
Carbon has 2 electrons in its first electron shell and in its valence electron shell (outer most shell) it has 4 electrons.
Carbon is unique in its ability to form a close to ten million different compounds. Carbon forms a stable structure with itself. Graphite is a Non-metallic, crystalline form. Diamonds are a Glossy, crystalline form.
Facts:
Ionization Energy: 11.260 eV
Oxidation States: +4, +2, -4
Electron Shell Configuration:
1s2
2s2 2p2
The slide show below is a series of images displaying carbons forms in their structures.